INTRODUCTION
Interstitial lung diseases (ILDs) describe a group of diseases that affect the lung parenchyma, characterized by different levels of inflammation, lung fibrosis, and gradual decline of respiratory functions1. They are now recognized to have systemic effects that go far beyond the lungs. Among those systemic effects, osteopenia and osteoporosis have been overlooked despite their clinical relevance2-4. Low bone mineral density (BMD) is one of the leading reasons for frailty, disability, and illness caused by fractures5. Some of the classical risk factors such as aging, smoking, chronic inflammation, and sedentary lifestyle are common symptoms, mirroring demographic and clinical profile of ILD patients5-7. Additionally, the specific pathophysiological processes of ILD, such as chronic hypoxemia, continuous inflammatory signaling, lack of proper nutrition, advancing physical incapacitation, and the steady use of corticosteroids, build a biological environment that may promote bone loss3,7.
Awareness of the burden of low BMD in ILD patients has critical implications for fracture prevention, improving quality of life, and addressing long-term outcomes in this vulnerable patient population6. The literature on this association is quite limited by factors such as small sample sizes, different types of ILD populations, and different BMD assessment methods7. While investigations suggest that reduced BMD may be common in idiopathic pulmonary fibrosis (IPF), connective tissue disease–associated ILD (CTD-ILD), and hypersensitivity pneumonitis, the evidence has never been synthesized systematically7. Thus, the literature lacks pooled estimates that could inform screening strategies, guide early intervention, or influence disease-specific management decisions.
This systematic review and meta-analysis aimed to address this gap by assessing the prevalence of low bone mineral density (osteopenia and osteoporosis) in adults with different ILD phenotypes within clinical settings.
METHODS
Data sources and search strategy
This systematic review and meta-analysis was conducted between October 2025 and January 2026, in accordance with the PRISMA 2020 guidelines. A comprehensive literature search was conducted in PubMed, Scopus, and Web of Science from inception to October 2025. The strategy combined controlled vocabulary and free-text terms covering two core concepts: 1) interstitial lung disease (including key ILD subtypes and CTD-ILD terms); and 2) low bone mineral density (osteopenia/osteoporosis and BMD/DXA-related terms). No language restrictions were applied. No filters for age, sex, setting, or study design were applied at the search stage. The full electronic search strategies for each database are provided in the Supplementary file.
Study selection criteria
Eligibility criteria were defined according to a PEO framework: Population=adults (aged ≥16 years) with a confirmed ILD diagnosis; Exposure=interstitial lung disease, including IPF, NSIP, hypersensitivity pneumonitis, fibrotic ILD, and CTD-ILD subtypes; Outcome=prevalence of osteopenia and/or osteoporosis based on BMD assessment (e.g. DXA).
Studies were eligible if they included adults (aged ≥16 years) with a confirmed ILD diagnosis (including IPF, NSIP, hypersensitivity pneumonitis, fibrotic ILD, and CTD-ILD subtypes) and used an observational design (cross-sectional, cohort with baseline data, registry/database/chart review, or clinical series with ≥10 ILD patients). Included studies were required to report, or allow calculation of, the prevalence of osteopenia and/or osteoporosis based on BMD assessment (e.g. DXA).
Studies were excluded if ILD data were not separable from non-ILD populations, if they were pediatric-only, if they lacked extractable prevalence data for osteopenia/osteoporosis, or if they were case reports/series with <10 ILD patients, reviews/editorials/letters without primary data, animal/in vitro studies, or studies primarily focused on malignancy-related bone disease.
Study screening
Title and abstract screening was performed independently by two reviewers using the predefined eligibility criteria. Articles passing this stage underwent full-text assessment by the same two reviewers. At each stage, disagreements were resolved through discussion, and where consensus could not be reached, a third reviewer was consulted. Reference lists of eligible articles were also screened to identify additional relevant studies
Data extraction
Two reviewers independently extracted data using a standardized form. Extracted items included: study characteristics (first author, year, country, design, and setting), ILD subtype and diagnostic approach, sample size and participant demographics (age, sex), relevant clinical variables (e.g. lung function where reported and glucocorticoid exposure), and BMD assessment method (e.g. DXA/DEXA or other techniques). For outcomes, we extracted the number of participants with osteopenia and/or osteoporosis, the denominator with valid BMD measurements, and calculated study-level prevalence when needed. Discrepancies were resolved by discussion.
Quality assessment
To evaluate the methodological quality of the 16 included studies, we applied the National Institutes of Health (NIH) quality assessment tool for observational cohort and cross-sectional studies. We examined each study’s internal validity by assessing key domains. Each article was ultimately rated as ‘good’, ‘fair’, or ‘poor’ based on its overall risk of bias and the clarity of its reporting. Items were scored: Yes=1 and No/CD/NR/NA=0, with Good=10–14, Fair=5–9, and Poor=0–4. The included case-control study was assessed using the NIH case-control tool and was rated Good (score 9). To ensure a consistent and objective review, any disagreements during the assessment were settled through a final group discussion (Supplementary file Table 1).
Statistical analysis
All analyses were performed in R statistical software (R Foundation for Statistical Computing, Vienna, Austria) using the meta package. The primary outcome was the prevalence of low bone mineral density (low BMD), calculated for each study as the proportion of affected participants among those assessed. Pooled prevalence was estimated using a random-effects generalized linear mixed model (GLMM) for proportions with a logit transformation (sm=‘PLOGIT’, method=‘GLMM’), with results back-transformed to the proportion scale. A continuity correction of 0.5 was applied to handle zero cells (incr=0.5; method.incr=‘all’). Between-study heterogeneity was evaluated using Cochran’s Q, I², and τ². Results were summarized using forest plots. Publication bias was assessed using funnel plots and formally tested using the Begg–Mazumdar rank correlation test and Egger’s regression test. Sensitivity analyses were conducted in R (e.g. influence/leave-one-out analyses) to assess the robustness of the pooled estimate.
RESULTS
Search results
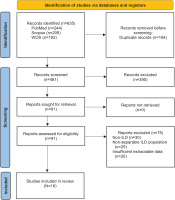
Our database search identified 635 records. After removing duplicates, 481 unique citations remained for title and abstract screening. Following this stage, 390 records were excluded for not meeting the eligibility criteria. Ninety-one articles underwent full-text review, and 75 were excluded because they did not meet inclusion criteria, mainly due to non-ILD, non-separable ILD populations, or insufficient extractable data on osteopenia/osteoporosis prevalence. Ultimately, 16 studies met all inclusion criteria and were included in the meta-analysis2,3,5,8-20. The PRISMA flowchart is shown in Figure 1.
Characteristics of included studies
Sixteen observational studies published between 2002 and 2025 were included, representing a geographically diverse distribution, with data originating from Europe (Italy, Spain, Switzerland, Greece), North America (USA, Mexico), and Asia (Saudi Arabia, Japan, India, Turkey) (6 studies from Europe5,10,13-15,20, 4 from North America3,9,11,17, and 6 from Asia2,8,12,16,18,19). Most studies used cross-sectional2,5,8-10,12,14,16 or retrospective designs3,11,15,19,20 and were conducted in tertiary/academic centers, including several study populations drawn from lung transplant evaluation centers (8 cross-sectional or cross-sectional case-control, 5 retrospective, and 3 prospective studies). The included populations covered a broad spectrum of ILD phenotypes: IPF-only studies, CTD-ILD/RA-ILD studies, hypersensitivity pneumonitis, occupational ILD (e.g. silicosis/pneumoconiosis), and less common ILDs such as lymphangioleiomyomatosis and pulmonary Langerhans cell histiocytosis, as well as mixed ILD/diffuse parenchymal lung disease studies. The demographic profile of the participants varied significantly according to the specific lung disease. Mean ages ranged from younger populations in the fourth decade of life (e.g. LAM and LCH studies) to elderly populations with a mean age reaching approximately 70 years in IPF and RA-ILD studies (reported mean ages ranged from 37.1 to 69.7 years). Gender distribution also varied widely; the dataset included female-only study populations (LAM and HP/CTD-ILD) and male-only study populations (silicosis and IPF), while 12 studies included mixed-sex populations3,5,8,10-15,18-20. Lung function data, where available, indicated moderate-to-severe disease, with mean forced vital capacity (FVC) %predicted values typically ranging between 51% and 88%.
Current or chronic glucocorticoid use was frequently reported across many studies, with prevalence rates reaching as high as 77% in some idiopathic pulmonary fibrosis (IPF) populations; glucocorticoid exposure was reported in 12 of the 16 studies2,3,5,8,9,11-13,15,16,18,20. Notably, two studies specifically recruited steroid-naive patients to evaluate the independent impact of interstitial lung disease (ILD) on bone mineral density (BMD). Regarding diagnostic tools, dual-energy X-ray absorptiometry (DXA) was the most frequently utilized method, although alternative approaches, such as computed tomography (CT) density scans of the thoracic vertebrae, were employed in one Japanese study12; 1 study used densitometry14 and 3 did not clearly report the BMD method8,10,19. The clinical burden of bone disease was substantial; the prevalence of osteoporosis reached as high as 44–51% in specific studies, while the total prevalence of low BMD, encompassing both osteopenia and osteoporosis, exceeded 80% in several tertiary and pre-transplant populations (Table 1).
Table 1
Characteristics of the included interstitial lung disease studies, including study design, setting, ILD phenotype, participant demographics, pulmonary function, glucocorticoid exposure, and bone mineral density assessment methods, 2002–2025 (16 studies)
| Authors Year | Country | Study design | Center type | Total ILD | ILD subgroup | Mean age (years) | Sex | FVC | Current GC use | BMD method | OPN/OPO/T | ILD with DXA |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Caffarelli et al.5 2016 | Italy | Cross-sectional | Specialized ILD clinic | 58 | IPF | 65.1±9.1 | M 39 F 19 | M 70.7±22.8 F 65.4±19.3 | 77% | DXA | 0/19/19 | 58 |
| Calmarza et al.15 2024 | Spain | Observational and retrospective study | General hospital | 38 | EPID | 60.3±5.8 | M 26 F 12 | 65 (IQR=16) | 50% (n=19) | DXA | 18/3/21 | 38 |
| Yıldızgören et al.16 2016 | Turkey | Cross-sectional | Training and research hospital | 104 | Silicosis/pneumoconiosis | NR | M 104 | NR | 0% (excluded) | DXA | 58/10/68 | 104 |
| Caplan-Shaw et al.3 2006 | USA (NY) | Retrospective cohort study | Tertiary care center | 86 | (DPLD), (UIP), (NSIP), and collagen vascular disease | 53±9 | M 49 F 37 | %pred 51±19 | 75% no-OP; 73% OP | DXA | 49/11/60 | 86 |
| Tschopp et al.13 2002 | Switzerland (Zurich) | Prospective data collection | University hospital | 74 | ILD/LT candidates | 39.2±11.7 | M 33 F 41 | NR | 60.8% (45/74) | DXA (Hologic) | 23/45/68 | 74 |
| Arenas-de Larriva et al.14 2010 | Spain (Cordoba) | Cross-sectional | University hospital | 156 | COPD, ILD, CF, miscellaneous | NR | M 109 F 47 | NR | NR | Densitometry | 22/15/37 | 55 |
| Lakey et al.11 2011 | USA (Durham, NC) | Retrospective study | University hospital | 137 | COPD, PF, and other diseases | 52.7±12.1 | M 74 F 63 | NR | 50% (chronic ≥3 m) | DXA (Hologic) | 67/51/118 | 137 |
| Makras et al.20 2011 | Greece (Athens and Thessaloniki) | Retrospective study | Single outpatient clinic | 25 | LCH | 37.1±2.7 | M 13 F 12 | NR | 0% | DXA (Lunar) | 2/2/5 | 25 |
| Taveira-DaSilva et al.17 2005 | USA (National Institutes of Health, Bethesda, Maryland) | Prospective cohort study | Specialized NIH center | 211 | LAM | 43.9±0.6 | F only | 3.09±0.04 L; 88.3±1.1% | NR | DXA | 100/49/149 | 211 |
| Alhamad et al.18 2015 | Saudi Arabia | Prospective study | ILD center | 196 | UIP, NSIP, sarcoidosis, chronic HP, cryptogenic OP | 58.1±14.5 | M 73 F 123 | FVC% no-OP 62.1±21.1; OP 64.4±18.6 | 43.4% (85/196) | DXA | 70/86/156 | 196 |
| Ikezoe et al.2 2015 | Japan | Cross-sectional | University hospital | 55 | IPF | 68±0.4 | M 55 | 88.1±21.3 | 0% | CT | 24/15/39 | 55 |
| Alhamad et al.19 2020 | Saudi Arabia | Retrospective review of an ongoing prospective database | Specialized ILD center | 212 | IPF | 66.4±11.2 | M 150 F 62 | 57.3±20 | NR | NR | 0/59/59 | 146 |
| Alarcón-Dionet et al.9 2024 | Mexico | Cross-sectional | Specialized ILD center | 50 | HP and CTD-ILD | 61±11 | F only | %pred 60±19 | 50% (5–10 mg/d) | DXA | 10/32/42 | 50 |
| Mena-Vázquez et al.10 2020 | Spain | Cross-sectional case-control study | Rheumatology clinics | 41 | CTD-ILD (RA-ILD) | 67.9±7.8 | M 21 F 20 | 72.1 (20.7) | NR | NR | 0/20/20 | 41 |
| Mochizuki et al.12 2025 | Japan | Cross-sectional | University hospital | 637 | ILD and COPD | 69.7±12.1 | M 120 F 517 | NR | 16.6% (n=106) | DXA | 0/0/64 | 97 |
| Aggarwal et al.8 2022 | India | Cross-sectional | Tertiary care hospital | 97 | Mixed ILD | 55.7±12.6 | F 61 | %pred OP 70.8±16.6; no-OP 68.9±15.8 | 37.1% (sig >10 mg, >3 m) | NR | 0/39/39 | 97 |
[i] ILD: interstitial lung disease. FVC: forced vital capacity. GC: glucocorticoids. BMD: bone mineral density. DXA/DEXA: dual-energy X-ray absorptiometry. QCT: quantitative computed tomography. CT: computed tomography. IQR: interquartile range. %pred, percent predicted. NR: not reported. IPF: idiopathic pulmonary fibrosis. FN: femoral neck. LS: lumbar spine. OPN: osteopenia. OPO: osteoporosis. OPN/OPO/T: osteopenia count/osteoporosis count/total low bone mineral density (osteopenia + osteoporosis). PE: prednisone equivalent. no-OP: no osteoporosis. OP: osteoporosis.
Quality was assessed using the NIH Quality Assessment Tool for observational cohort and cross-sectional studies as presented in Supplementary file Table 2.
Overall pooled prevalence of low BMD in interstitial lung disease
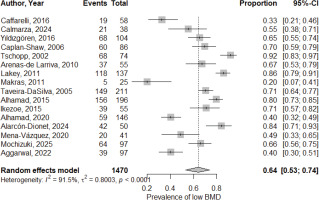
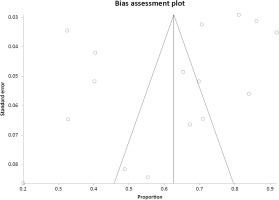
Using a random-effects GLMM with logit transformation, the pooled prevalence of low BMD among patients with ILD was 0.64 (64%; 95% CI: 0.53–0.74; total n=1470), with high heterogeneity (I2=91.5%, τ2=0.8003, p<0.0001) as shown in the forest plot (Figure 2). The funnel plot (Figure 3) is shown, and small-study effects were assessed using the Begg–Mazumdar test (Kendall’s τ= -0.283333, p=0.116) and Egger’s test (bias= -5.718144; 95% CI: -13.128106–1.691817, p=0.1201).
Subgroup analysis
Subgroup analyses demonstrated that osteopenia and osteoporosis were both frequent among ILD populations, with pooled prevalence of 39% (95% CI: 32–48; 11 studies) and 31% (95% CI: 22–41; 15 studies), respectively, and substantial between-study heterogeneity for each outcome (I²=78.9% and 88.2%) was observed. Tests for small-study effects for these outcome-specific analyses (Begg–Mazumdar and Egger) are summarized in Table 2.
Table. 2
Outcome-specific pooled prevalence of osteopenia and osteoporosis among adults with interstitial lung disease, with small-study effect assessments (N=1470; 16 studies)
When low BMD (osteopenia and/or osteoporosis) was examined across clinical strata, prevalence estimates varied by disease context and patient characteristics. The highest pooled prevalence was observed in pre-transplant/end-stage studies (77%; 95% CI 63–87; 5 studies), while elevated prevalence was also seen in studies with high glucocorticoid exposure (74%; 95% CI: 55–87; 6 studies). Among sex-restricted studies, pooled prevalence was 75% (95% CI: 63–84; 2 studies) in female-only studies and 67% (95% CI: 60–74; 2 studies) in male-only studies. By ILD phenotype, pooled prevalence was 57% (95% CI: 46–67; 2 studies) in CTD-ILD and 48% (95% CI 30–66; 3 studies) in idiopathic fibrotic ILD. Prevalence also remained high across lung function strata, including 62% (95% CI: 45–77; 2 studies) for FVC 50–69% and 58% (95% CI: 44–71; 2 studies) for FVC ≥70% (Table 3).
Table 3
Subgroup analyses of pooled prevalence of low bone mineral density across clinical and disease-specific strata in interstitial lung disease studies, 2002–2025 (16 studies)
Sensitivity analysis
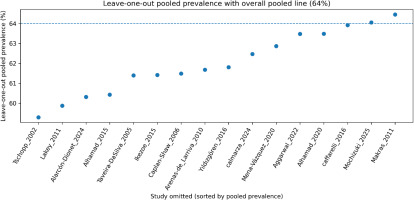
Leave-one-out sensitivity analysis (Figure 3) showed that the pooled estimate was stable: after omitting each study in turn, the pooled prevalence ranged from 59.3% to 64.5% (maximum absolute change ≤2.7 percentage points). The largest decrease occurred when Tschopp et al.13 was omitted (59.3%), and the largest increase when Makras et al.20 was omitted (64.5%). Heterogeneity remained high across all iterations (I² ~ 92.2–94.2%), indicating that no single study drove the overall findings.
DISCUSSION
In this systematic review and meta-analysis, we demonstrate that low BMD is highly prevalent among adults with ILD, affecting nearly two-thirds of patients. Both osteopenia and osteoporosis were common, with pooled prevalence estimates of 40% and 31%, respectively. Importantly, the burden of bone disease was not confined to a single ILD subtype or clinical context but was consistently observed across disease phenotypes, sexes, lung function strata, and geographical regions. These findings position low BMD as one of the most frequent and clinically relevant extrapulmonary comorbidities in ILD.
Our pooled prevalence of 64% for low BMD substantially exceeds estimates reported in age-matched general populations worldwide, where osteoporosis prevalence typically ranges between 10% and 25% depending on sex and region21. Even when compared with other chronic respiratory diseases, such as chronic obstructive pulmonary disease (COPD), the burden observed in ILD appears comparable or higher, particularly in advanced disease stages22. These findings suggest that ILD confers disease-specific risks for skeletal fragility that extend beyond shared demographic and lifestyle risk factors.
The consistently high prevalence across diverse ILD phenotypes underscores that bone loss is not restricted to a single pathogenic pathway but rather represents a convergent consequence of chronic lung disease, systemic inflammation, and progressive functional impairment. Several interrelated mechanisms probably account for the high prevalence of low BMD reported in ILD populations. Hallmarked by chronic systemic inflammation, many ILDs activate osteoclasts by signaling them with pro-inflammatory cytokines, e.g. tumor necrosis factor-α, interleukin-1, and interleukin-6, thereby resulting in increased bone resorption23,24. On the other hand, fibrotic ILDs, namely idiopathic pulmonary fibrosis (IPF) and connective tissue disease-associated ILD (CTD-ILD), have additionally been marked by a dysregulated transforming growth factor-β signaling, which is known to be a pivotal factor in the pathological process of abnormal bone remodeling.
Chronic hypoxemia of moderate-to-advanced ILD patients may thus be an additional factor for osteoblast impairment and disruption of calcium metabolism25-27. At the same time, the patients’ reduced physical activity caused by dyspnea, fatigue, and exercise intolerance, leads to the mechanical unloading of bones, resulting in the acceleratory phenomenon of bone loss28. The shortage of nutrients such as vitamin D and proteins and the presence of hypoalbuminemia, which are commonly found in the ILD population, are other factors that can make the bone more vulnerable29-30. Importantly, these disease-intrinsic mechanisms may work regardless of corticosteroid exposure, as illustrated by studies conducted on steroid-naïve ILD populations that still showed decreased BMD. This reinforces the argument that ILD per se is an independent risk factor state for osteoporosis2,16,20.
The highest pooled prevalence of low BMD was observed in pre-transplant and end-stage ILD studies (77%). This finding is biologically plausible and clinically expected, as these patients represent the extreme end of disease severity, with prolonged immobility, advanced hypoxemia, systemic inflammation, and cumulative exposure to medications that adversely affect bone health11,31. Given the dramatic consequences of fractures in transplant candidates, including delayed listing, perioperative complications, and increased mortality, these data reinforce the need for aggressive bone health surveillance in this subgroup11,31,32.
Steroid exposure was another major factor of skeletal burden. Patients from studies with high steroid use demonstrated markedly elevated prevalence estimates (74%), consistent with the well-established effects of corticosteroids on osteoblast suppression, osteocyte apoptosis, and increased fracture risk32,33. Notably, however, low BMD was also common in studies without glucocorticoid exposure, emphasizing that steroid therapy amplifies, but does not solely account for, bone loss in ILD.
On gender-restricted analyses, very high prevalence rates were observed in both female-only (75%) and male-only (67%) studies. Postmenopausal women suffer from bone loss due to estrogen deficiency21 but the large quantity of bone loss documented in males-only studies2,16 puts into question the traditional view of osteoporosis as a primarily female condition and points to the need of screening men with ILD, particularly those living with fibrotic disease or those having occupational exposure, more often.
Across ILD subtypes, CTD-ILD showed a higher pooled prevalence (57%) compared with idiopathic fibrotic ILD (48%). This difference likely reflects the additive effects of systemic autoimmune inflammation, glucocorticoid and immunosuppressive therapy, and multisystem involvement inherent to connective tissue diseases. Orphan ILDs demonstrated wide confidence intervals, reflecting both biological heterogeneity and limited data, underscoring the need for disease-specific studies in these rare populations, suggesting that further disease-specific studies in these rare populations would be informative18.
Interestingly, low BMD prevalence remained high across lung function strata, including patients with relatively preserved FVC. This suggests that skeletal involvement may develop early in the disease course and is not solely a sequela of advanced physiological impairment, aligning with prior observations that bone loss may precede overt respiratory disability34. Previous individual studies have reported highly variable prevalence estimates of osteoporosis in ILD, ranging from as low as 13% to over 50%, largely due to differences in study design, patient selection, disease severity, and BMD assessment methods2,3,5,8,18. By pooling data across diverse studies and applying robust statistical modeling, our study provides the most comprehensive estimate to date and reconciles much of this apparent inconsistency.
A persistent controversy in the literature concerns whether reduced BMD in ILD is driven primarily by disease severity, chronicity, or treatment exposure3,5,7. Our findings suggest that all three contribute, but none alone fully explain the observed burden. The persistence of high prevalence in steroid-naïve and milder disease studies supports a disease-intrinsic effect, while the marked elevation in advanced and heavily treated populations highlights cumulative risk.
The high prevalence of low BMD demonstrated in this meta-analysis has important clinical implications. Fractures in ILD patients may lead to rapid functional decline, impaired respiratory mechanics, increased hospitalization, and excess mortality35,36. Despite this, bone health assessment is not routinely integrated into ILD management pathways or clinical guidelines. Our findings support the incorporation of routine BMD screening into standard ILD care, particularly for patients with fibrotic disease, CTD-ILD, glucocorticoid exposure, or advanced disease. Early identification of osteopenia provides an opportunity for timely intervention through lifestyle modification, nutritional optimization, vitamin D and calcium supplementation, and pharmacological therapy when appropriate. Recognizing osteoporosis as a core extrapulmonary manifestation of ILD may improve holistic disease management and quality of life, aligning ILD care with multidisciplinary, patient-centered models increasingly advocated in chronic respiratory medicine.
Strengths and limitations
This study has several strengths, including comprehensive coverage, rigorous methodology, use of a GLMM approach for prevalence estimation, and extensive subgroup and sensitivity analyses demonstrating the robustness of our findings. To our knowledge, this is a novel meta-analysis to quantify the burden of low BMD across ILD phenotypes and clinical contexts. Nevertheless, several limitations merit consideration. First, substantial heterogeneity was present across analyses, reflecting true clinical diversity as well as differences in study design, populations, and BMD assessment methods. Second, most included studies were observational and cross-sectional, limiting causal inference and precluding assessment of fracture outcomes. Third, detailed data on vitamin D status, physical activity, cumulative steroid dose, and fracture incidence were inconsistently reported, preventing more granular analyses. Additionally, BMD classification based on DXA relies on reference population standards (e.g. WHO T-score criteria), and differences between WHO and local or population-specific reference databases may influence the classification of osteopenia and osteoporosis, potentially affecting prevalence estimates across studies in different geographical and ethnic settings. Future research should focus on prospective longitudinal studies to clarify temporal relationships between ILD progression and bone loss, identify high-risk phenotypes, and evaluate fracture incidence and outcomes. Interventional trials assessing targeted bone-protective strategies in ILD populations are also warranted. Standardization of BMD assessment methods and reporting would further enhance comparability across studies.
CONCLUSIONS
This meta-analysis shows that low bone mineral density constitutes a significant burden among patients with interstitial lung disease. Osteopenia and osteoporosis were frequently observed across various ILD populations, and the burden was highest in pre-transplant individuals and among those with high glucocorticoid exposure. However, substantial heterogeneity existed across studies. These findings suggest that routine bone health assessment and preventive strategies may be an important consideration in ILD care. Future well-designed prospective studies are needed to clarify mechanisms, standardize evaluation, and evaluate targeted interventions.